Everything in our world is built from the smallest units of matter: atoms. These building blocks are composed of three subatomic particles: protons, neutrons, and electrons. Together, these particles define the properties of a specific element.
An isotope is a variation of a certain element. Isotopes of the same element share the same number of protons and the same arrangement of electrons, but the number of neutrons differs. Due to this difference the physical properties of isotopes vary. For example, some isotopes are unstable, and others are not.
To regain stability, unstable isotopes shed excess particles and energy in the form of radiation and are therefore called radioactive isotopes. These radioisotopes are also referred to as atoms with too much energy in their nucleus, or with an unstable combination of neutrons and protons.
Send us an email

Radioisotopes can occur naturally or are synthesised by altering the atom. Some elements have many different kinds of isotopes. For example: the element iodine has two isotopes that occur in nature (I-127 and I-129), and 35 others are artificially created. Four of these (I-123, I-124, I-125, and I-131) are used in radiotherapy and clinical imaging such as SPECT or PET.
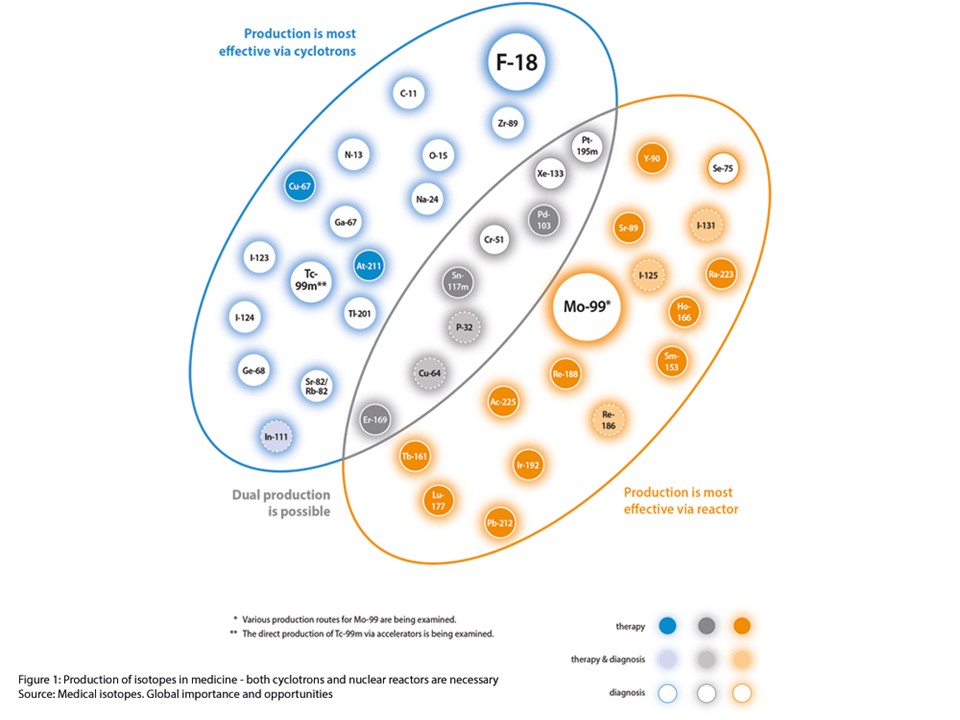
Radioisotopes are either produced in a nuclear reactor or cyclotron. Neutron-rich radioisotopes, such as molybdenum-99, are best produced in a nuclear reactor, whereas proton-rich radioisotopes are generated in a cyclotron, also known as a particle accelerator. Generally speaking, cyclotron isotopes are short-lived nuclides with only several hours of half-life. Reactor isotopes usually have relatively longer half-lives allowing for a longer, global geographic reach. The production route is determined depending on the application.
A nuclear reactor, such as the High Flux Reactor (HFR) in Petten, the Netherlands, irradiates atoms to make them radioactive, thus producing a radioisotope. This irradiated material is then turned into a radiochemical, which is the basic material used in nuclear medicine. More details about the production of medical isotopes and the supply chain of nuclear medicine can be found on the NRG website.
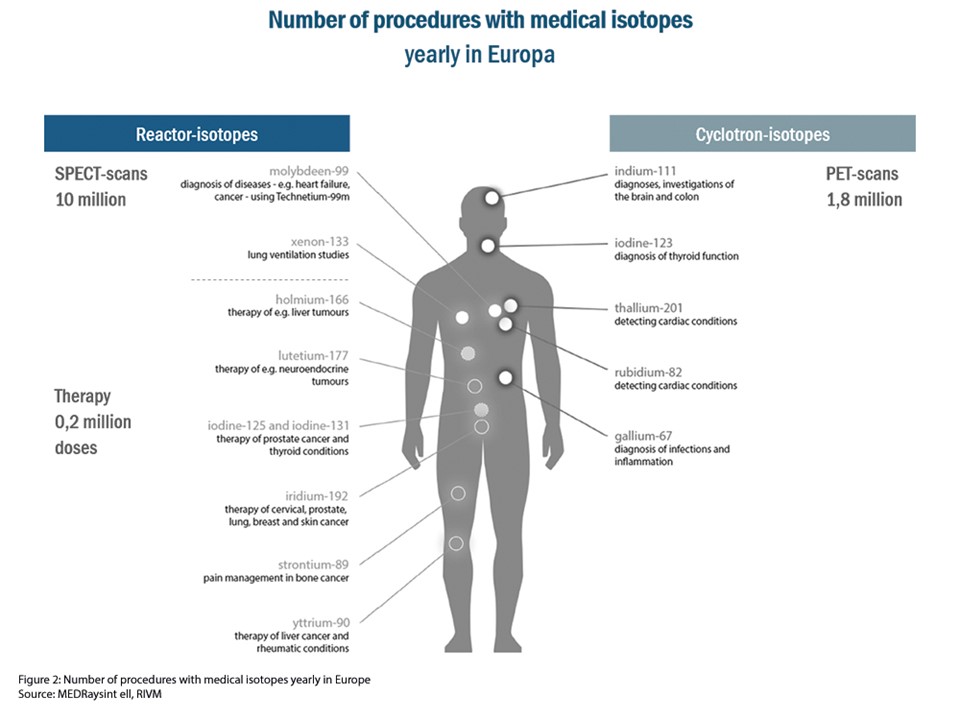
Isotopes are an essential part of radiopharmaceuticals, which are used to detect (diagnosis) and treat (therapy) cardiovascular diseases and cancer. Each year 48 million patients are diagnosed or treated with these nuclear medicines. Every day more than 30,000 people worldwide are helped with medical isotopes that are produced in the High Flux Reactor (HFR) in The Netherlands.
Radionuclides, another word for isotopes used in medicine, facilitate imaging. Nuclear physicians use them to diagnose the disease and the stage it is in by injecting the patient with a small amount of radioactive fluid. A SPECT or PET scanner detects the radiation that is left behind in the patient’s tissue and creates an image that allows the nuclear physician to see how an organ, such as the heart, the liver, or the brain, is functioning or whether there are active cancer cells.
And after therapy, nuclear specialists can monitor the response to the treatment by using the radiation that medical isotopes transmit.
Isotopes in medicine are used to kill cancer cells. External radiation therapy (teletherapy) is one way of using radioisotopes. The main challenge with this kind of nuclear therapy is that it can cause damage to healthy tissue as well. To minimise this hazard, scientists are continuously finding new ways for personalised treatment: treatment of diseases that is adapted to an individual and her or his specific disease. This leads to a more effective treatment with fewer side effects. Radionuclides play an important role in this development.
An example of personalised treatment with radionuclides is personalised, local therapy, also known as short-range radiotherapy, such as brachytherapy (see below). The isotopes best suited for this kind of nuclear medicine are alpha or beta emitters, which release their radiation close to their targets.
Targeted radionuclide therapy
Another example of personalised, local therapy is targeted radionuclide therapy, during which a radioactive substance is administered to the patient. The nuclear substance travels through the bloodstream and only targets cancer cells by binding with the tumour and its metastases. It kills the diseased cells by damaging their DNA. The targeting molecules (also known as tracers) that transport medical isotopes to the cancer cells are chosen by their affinity to the cell’s target structures, such as receptors or antigens. Collateral damage to other, healthy cells is therefore minimised.
Therapy with nuclear medicine: